Host Cell Proteins
In der biopharmazeutischen Herstellung werden unter dem Begriff Host Cell Proteins (HCPs) Proteinverunreinigungen aus dem Wirtsorganismus zusammengefasst, die während des Herstellungsprozesses von monoklonalen Antikörpern, Antikörper-Wirkstoff-Konjugaten, therapeutischen Proteinen und anderen Biopharmazeutika auf Proteinbasis auftreten.
Was sind Host Cell Proteins?
Host Cell Proteins sind Verunreinigungen aus dem Wirtsorganismus, die bei der Herstellung von Biopharmazeutika verwendet werden. Diese Proteine können während des Herstellungsprozesses zusammen mit dem gewünschten therapeutischen Protein aufgereinigt werden. Sie sind unerwünschtes Nebenprodukt da sie die Sicherheit, Wirksamkeit und Stabilität des Endprodukts beeinträchtigen können.
Die Quantifizierung und Entfernung von HCPs im endgültigen Arzneimittelprodukt auf akzeptable niedrige Werte ist unerlässlich. Verunreinigungen, die mit rekombinanten Biotherapeutika einhergehen, können die Wirksamkeit des Medikaments erheblich beeinträchtigen und Immunogenität verursachen.
HCP Immunoassays
Eine der wirksamsten Methoden zur Identifizierung und Isolierung von Wirtszellproteinen ist ein HCP-Immunoassay. Dieser spezialisierte Test zielt auf alle Proteine ab, die von der Zelllinie produziert werden, die bei der Herstellung des therapeutischen Biopharmazeutikums verwendet wird, und zwar mit einem Antikörper, der gegen alle oder so viele Proteine wie möglich gebildet wird. Während des Bioprozesses mit mehreren verschiedenen Schritten werden Anti-Wirtszellprotein-Immunoassay-Proben entnommen und zur Messung des Gesamtgehalts an HCP und zur Festlegung von Spezifikationen für diesen Bioprozess verwendet.

Custom HCP Antikörper- und Assays
Custom HCP Antikörper- und Assays
Profitieren Sie von der Erfahrung aus zwei Generationen wissenschaftlicher Fortschritte. Rockland ermöglicht seit 1962 die Detektion, Charakterisierung und HCP-Analyse von Biomolekülen.
HCP Antibodies HCP Assay Development
Profitieren Sie von der Erfahrung aus zwei Generationen wissenschaftlicher Fortschritte. Rockland ermöglicht seit 1962 die Detektion, Charakterisierung und HCP-Analyse von Biomolekülen
HCP Antibodies HCP Assay DevelopmentCustom HCP Antikörper- und Assays
Sind Sie an einem maßgeschneiderten Antikörper und Assays interessiert, die auf Ihre Bedürfnisse zugeschnitten sind, um Ihren biologischen Produktionsprozess auf HCP-Verunreinigungen zu untersuchen? Dann haben wir das richtige Programm für Sie parat.
Profitieren Sie von der kombinierten Expertise von antibodies-online und unserem Partner Rockland Immunochemicals, Inc. (Rockland). Nutzen Sie die mehr als 15-jährige Erfahrung von Rockland bei der Herstellung von maßgeschneiderten polyklonalen HCP-Antikörpern, damit Ihr biopharmazeutisches Entwicklungsprogramm termingerecht und effizient verläuft.
Unsere umfassenden, vollständig anpassbaren Dienstleistungen decken alle Aspekte der Herstellung von Anti-HCP-Antikörpern ab, von der Antigenpräparation und Assay-Entwicklung bis hin zur Dokumentation und Herstellung.
Verschiedene Expressionswirtszellen erfordern unterschiedliche HCP-Produkte
Die Wahl des Wirts hat den größten Einfluss auf die Anzahl der unerwünschten Proteine. E. coli hat zum Beispiel nur ~4.300 Gene, während CHO-Zellen ~30.000 Gene haben. Zwar wird nicht jedes Wirtsgen transkribiert und in ein Protein übersetzt, aber die Komplexität des Wirtsgenoms zusammen mit den posttranslationalen Modifikationen in Säugetierzellen macht die Reinigung von Wirtszellverunreinigungen noch komplexer.
Die am häufigsten verwendete Säugetierzelllinie für die Herstellung therapeutischer monoklonaler Antikörper und anderer biologischer Arzneimittel sind CHO-Zellen. Einer der großen Vorteile von CHO-Zellen ist, dass sie große Mengen rekombinanter Proteine produzieren (3-10 Gramm pro Liter Kultur). Ein weiterer großer Vorteil sind die posttranslationalen Modifikationen der rekombinanten Proteine, die bei menschlichen Anwendungen sehr gut funktionieren.
Auf der anderen Seite bringt die Wahl von CHO als Expressionssystem auch Schwierigkeiten mit sich, insbesondere im Hinblick auf die Menge der Wirtszellproteine. E. coli hat nur ~4.300 Gene, während CHO-Zellen ~30.000 Gene haben. E. coli hat beispielsweise nur ~4.300 Gene, während CHO-Zellen ~30.000 Gene haben.3 Obwohl nicht jedes Wirtsgen transkribiert und in ein Protein übersetzt wird, trägt die Komplexität des Wirtsgenoms zusammen mit den posttranslationalen Modifikationen, die in Säugetierzellen vorhanden sind, zu einer weiteren Komplexität bei der Reinigung von Wirtszellverunreinigungen bei. Daher ist die Überwachung und Verringerung der aus CHO-Zellen stammenden HCPs eine der größten Herausforderungen im Produktionsprozess biologischer Arzneimittel.
HCP Antikörper, Kits und Proteine
Nachstehend finden Sie eine Übersicht der von antibodies-online.de angebotenen HCP-Antikörper, -Kits und -Proteine. Derzeit sind Antikörper mit der Reaktivität E. coli, CHO und HEK 293T verfügbar. Weitere Informationen erhalten Sie, wenn Sie auf die Links in der Tabelle klicken. Sie können den HCP-Antikörper, den Sie benötigen, nicht in unserer Liste finden? Wenden Sie sich an unser Expertenteam, um maßgeschneiderte HCP-Lösungen für Ihren Bedarf zu besprechen.Produkt im Fokus: AccuSignal™ E. coli HCP ELISA Kit (ABIN7542932)
Rocklands AccuSignal™ E. coli HCP ELISA Kit ist ein robuster Sandwich-Enzymimmunoassay (ELISA), der entwickelt wurde, um kontaminierende E. coli-Wirtszellproteine (HCPs) in therapeutischen Produkten während des gesamten Reinigungsprozesses nachzuweisen – sowohl in frühen, mittleren, späten als auch in finalen Stadien. Zusätzlich können HCPs der beiden häufig in der Bioproduktion von Arzneistoffen eingesetzten E. coli-Stämme B und K-12 erkannt werden.
E. coli-HCPs aus einer Probe werden auf der Platte des AccuSignal™ E. coli HCP ELISA Kits gebunden, auf der anti-E. coli-HCP-Antikörper bereits immobilisiert sind. Die Detektionsantikörper des Kits sind mit Meerrettichperoxidase (HRP) konjugiert. Beim Kontakt mit dem Substrat 3,3’,5,5’-Tetramethylbenzidin (TMB) aktiviert HRP eine Farbreaktion, deren Intensität mittels eines Plattenlesegeräts bei 450 nm gemessen werden kann. Dieser optische Dichtewert (OD) ist direkt proportional zur Konzentration der E. coli-HCPs, welche durch Interpolation anhand einer Vier-Parameter-Logistischen (4PL) Standardkalibrationskurve ermittelt werden kann.
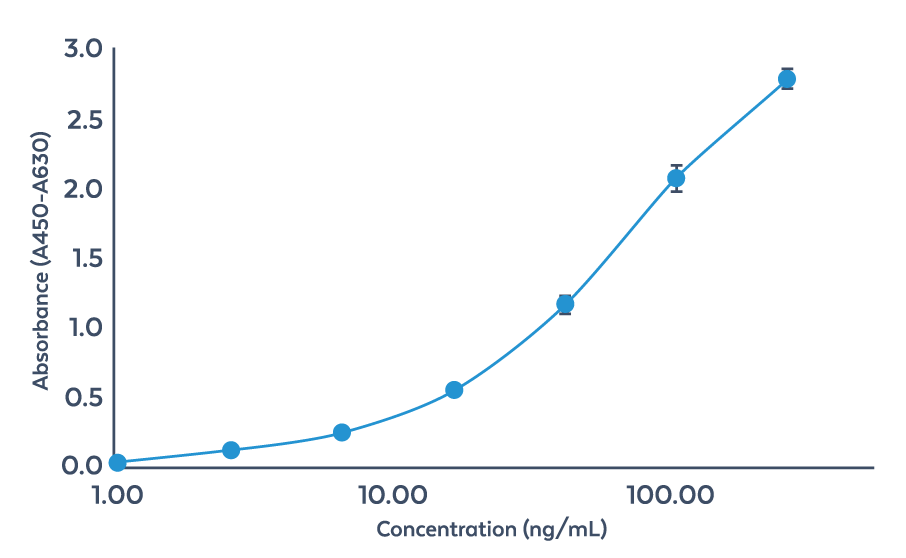
Vier-Parameter-Logistische (4PL) Standardkalibrationskurve für ABIN7542932
Weitere HCP Kits
HCP Antikörper
- (1)
- (2)
- (3)
- (1)
- (2)
- (2)
- (2)
- (2)
- (2)
HCP Proteine
- (2)
Referenzen
- : "Production of recombinant protein therapeutics in cultivated mammalian cells." in: Nature biotechnology, Vol. 22, Issue 11, pp. 1393-8, (2005) (PubMed).
- : "The art of CHO cell engineering: A comprehensive retrospect and future perspectives." in: Biotechnology advances, Vol. 33, Issue 8, pp. 1878-96, (2016) (PubMed).
- : "Host cell proteins in biologics development: Identification, quantitation and risk assessment." in: Biotechnology and bioengineering, Vol. 103, Issue 3, pp. 446-58, (2009) (PubMed).
- : "Profiling of host cell proteins by two-dimensional difference gel electrophoresis (2D-DIGE): Implications for downstream process development." in: Biotechnology and bioengineering, Vol. 105, Issue 2, pp. 306-16, (2010) (PubMed).
- : "Host Cell Proteins in Biologics Manufacturing: The Good, the Bad, and the Ugly." in: Antibodies (Basel, Switzerland), Vol. 6, Issue 3, (2017) (PubMed).
- : "The future of host cell protein (HCP) identification during process development and manufacturing linked to a risk-based management for their control." in: Biotechnology and bioengineering, Vol. 112, Issue 9, pp. 1727-37, (2016) (PubMed).
- : "Comprehensive assessment of host cell protein expression after extended culture and bioreactor production of CHO cell lines." in: Biotechnology and bioengineering, (2022) (PubMed).
- : "Towards continuous mAb purification: Clearance of host cell proteins from CHO cell culture harvests via "flow-through affinity chromatography" using peptide-based adsorbents." in: Biotechnology and bioengineering, Vol. 119, Issue 7, pp. 1873-1889, (2022) (PubMed).
Transcripts:
What is a host cell protein and why is it important in biopharmaceutical manufacturing?
A host cell protein is a contaminant that comes from manufacturing of biopharmaceuticals. Biopharmaceuticals are made using an engineered cell line that produces a therapeutic protein. Often times that can be a therapeutic monoclonal antibody. One of the major issues in the purification of the therapeutic monoclonal are proteins that are contaminating coming from the host cell line.
What is a host cell protein immunoassay and what is it used to monitor?
A HCP immunoassay is a specialized assay that is against all of the proteins of the cell line that is used to manufacture the therapeutic biopharmaceutical. It is a very complicated assay where there is an antibody reagent generated against all of the proteins or as many of the proteins as can be generated against and it is used to monitor in-process consistency in the purification of that molecule. What will happen is during the bioprocessing with multiple different steps samples are be taken in this anti-host cell protein immunoassay will be used to measure the level of total HCP present and it is used to set specifications for that bioprocess.
How to select the upstream HCP reference sample? Is it appropriate to take a single upstream HCP sample or should a downstream be used as well?
There is a couple different ways to do that. The reference sample should be chosen, should be representative of your upstream bioprocessing step. Generally it would be considered to be a crude fraction. Maybe sometimes initially post-harvest, but not too far into the bioprocessing workflow.
The second part of the question which was whether or not a single upstream sample was suitable or something also downstream that might be more representative of the enriched contaminating host cell proteins is appropriate, and there is not a single answer for that question. If the resources are available, it is probably good to do both.
Upstream reference materials are going to capture more of the contaminants and it is a good representation of what is happening in the bioprocessing and if there is any issue with the bioprocessing workflow.
Having a downstream sample can be useful for detecting those more specific host cell proteins that may become problematic as the material becomes more pure during its bioprocessing workflow.
What are the animal species used for making HCP reagents?
The animal species used for making anti-host cell protein reagents, most commonly is rabbit or goat. They have been used historically in almost equal amounts. Both are suitable for generating a good broad coverage against all of the HCP proteins, where broad coverage is defined as covering all molecular weights from the very small proteins to the very large proteins.
Typically for the antibodies multiple different animals are used to increase the diversity of the immune response, because any single animal alone will not make antibodies against all of the proteins that are required. So having a diversity of numbers of animals of either species is very important. And then in certain cases – again, if resources are available – one could make reagents using both species and then have the option to pick the one that worked best, or provided the best coverage against the host cell proteins.
It is worth to mention that another species is now being used more frequently. That is chicken. The rational for using chicken is, that it is evolutionarily more diverse or unrelated to the mammalian cells that are used to produce the biopharmaceuticals and it offers the ability to create a more robust immune response against more targets. It hasn’t been used to loo long at this time, so it is difficult to say whether or not, that is true.
What are the ways to improve or ensure good coverage of the produced anti-HCP reagent?
Generating anti-host cell protein reagents can be a challenging process. There are certain protein species within the sample that are poly-immunogenic or at low abundance and often times do not give a good immune response when analyzing by western blot. And so there are a couple of different ways to approach resolving that problem.
One of them is to fractionate the protein into high and low molecular weight species and then immunize one of your animal cohorts with the low molecular weight fraction. What that can do is give you an enhanced immune response or enriched immune response against those low molecular proteins specifically. That can then be combined back with the antisera that was generated against the total HCP reference sample.
Another thing that has been done is a technique called cascade immunization. Cascade immunization is one whereby an animal cohort is immunized with the host cell proteins. Frequently, what will happen is there is several immuno-dominant species of proteins that will demonstrate very good reactivity in western blots. What can be done is they can be purified and used to make an affinity matrix. That affinity matrix can be then used to deplete the host cell protein reference standard of those immuno-dominant protein species. And then the left over material can be concentrated and reimmunized. That can be done a few times and what happens over time – when it works right – is that you start to develop an immune response to the poorly immunogenic proteins.
What techniques are used to qualify HCP antibodies?
2D western blot or DIGE is a technique where the HCP proteins are separated based on their isoelectric point and molecular weight. That’s important because there are so many protein species present in the sample that a simple 1D western blot is not sufficient to see whether or not you are detecting all the protein which maybe masked under the other proteins. So, the way to approach that problem is to separate them using this 2-dimensional technique. They can be measured by western blot where you are demonstrating how many of the existing host cell proteins present in the sample can be detected by an antibody.
And then the DIGE technique, very similar to western blot, uses a fluorescence label and can also be used to determine how many of the HCP protein species are detected by the antibody. The coverage component is really an arbitrary measurement and it is meant to be an indication of the quality of the coverage. General ranges can be 60 to 80% and the number really needs to be a demonstration of the coverage across the whole entire blood. So it can’t just come from one single area.
So as long as your coverage is across most of the protein species over all ranges of isoelectric points and molecular weights and your coverage is above 60 or 80% the antibody is probably a good reagent.
How is an antisera pool made?
Frequently, when producing an anti-host cell protein antibody multiple different animals will be in a cohort for generation of antisera. Not all of the animals are going to be responding in the way that is needed to produce the best possible reagent. Frequently a selection of a subset of the sera collected from those animals will be used. So, the way that that is done: analytical testing is done, that can be 1D western blot and ultimately 2D western blot or DIGE experiments to demonstrate that the reagents have good coverage.
Those reagents that are sufficiently good to get you into the 60 to 80% coverage range across most of the different types of protein species can be pooled together to create a super pool. That then creates a single lot of antisera that can be used for downstream of all of the other things that are needed in purification and assay development.
How is mass spec being used as an orthogonal method for HCP analytical work?
Recently an orthogonal method has been added in addition to 2D western blot as a way to validate anti-host cell protein antibodies. And mass spec has some advantages in that it does not require immunogenicity of the protein species in order to detect it. So in that sense it is unbiased at the different anti-host cell protein species.
What it is been used for frequently now is to characterize the upstream HCP harvest sample in the bioprocessing stream to identify what are all the species that are in there. Subsequently it is now also used to validate anti-host cell protein reagent to complement the 2D western blot experiment and say what specifically is in the sample and also verify that the coverage is acceptable and good.
What is the best method for purification of the HCP-antibody reagent? Purification by Protein A or G, or affinity purification?
A frequent question that often comes up about anti-host cell protein antibodies is what to do once the antisera is generated and qualified. There’s a couple of different decisions to be made and there’s a lot of arguments in the field about what’s the best choice to make.
One is protein A or protein G purification which is used to isolate the IgG fraction of the anti-host cell protein antibody and the other purification method is affinity purification against an HCP reference standard affinity matrix. There is positives and negatives to both and neither one of these procedures is thought to be superior to the other. Protein A has the advantage of being unbiased in its selection of the antibodies. The disadvantage of protein A is that it does not enrich for your anti-host cell protein antibodies but also carries over all of the non-specific antibodies and can lower sensitivity of an immunoassay during assay development.
The advantages of affinity purification are that it can enrich for anti-host cell protein antibodies. So most of the things are going to be reactive in your final antibody purified material are going to be reacting to your anti-host cell proteins. It has the potential problem of being very biased towards the highly immunogenic proteins and may not purify equally across all different versions of antibodies for all the host cell proteins.
So, it is really a kind of a choice of the end user what they think might work best for them, because both have positives and negatives and both are acceptable methods for creating a reagent when submitting to the regulatory agencies.
Does the HCP antibody reagent support a single process or platform process? If platform, how many products can it support?
The anti-host sell protein antibodies can be used to support single processes. In the case where there is a new product being made it requires a special process. In that instance it may be able to use a generic or a custom HCP reagent. And in that case the reagent can be made and tested.
The point here about single process versus platform that’s important to know, if the reagent is going to support platform it is very important that a large amount of material be made to support the platform as it is unknown – generally when it is first started – how many different programs it is going to support. That means that if it starts with three drug programs on that platform it is pretty easy to determine how much of the anti-host cell protein antibody you will need to support that over the long term, however if that platform expands out to five or six or more drug programs you can very quickly deplete that antibody material.
So it is a really important in decision making about how much material you need to make for the long term, to make sure you don’t run out of critical material. Remanufacture, requalifying can be a time consuming and burdensome process that no one wants to go through.

Creative mind of antibodies-online with a keen eye for details. Proficient in the field of life-science with a passion for plant biotechnology and clinical study design. Responsible for illustrated and written content at antibodies-online as well as supervision of the antibodies-online scholarship program.
Zur Autorenseite



